Charm Sciences innovates and develops food safety and sanitation diagnostic solutions to predict and prevent chemical and microbiological challenges in farm-to-table food production and hospitality applications. We are committed to protecting our customers’ brands and helping them provide the highest quality products and services for our families.
Trusted
We are trusted by governments, organizations, and consumers around the world. We’re trusted because we’re completely focused on the betterment of the industry. We share innovation, best practices, training, and technology with our customers in an effort to further global—and local—best-in-class food safety programs.
Accurate
Our food safety customers trust our solutions because they are 100% accurate, easy to use, and tailored to be the exact fit for their needs. In fact, because of our commitment to food safety, never in the history of Charm, has a single Charm customer had to manage a product recall while actively—and properly—using our solutions.
Committed
Founded in 1978, Charm Sciences has made the science of food safety a way of life. From source to global supply chain to refrigerator, Charm is committed to developing, delivering, and supporting the very best in safety practices, technology and solutions for the dairy, food, and grain industries.
From Massachusetts to Australia, Charm Sciences Provides End-to-End Food Safety Solutions
DAIRY
The issues facing the global dairy industry, coupled with an ever-shifting landscape demand that you use an agile and accurate testing methodology, tests, and equipment to meet them. Over 75% of the global raw milk supply is tested with Charm products. Wherever you’re located, whether you’re testing cow, goat, sheep, or camel-based dairy products, we’ve got you covered.
FOOD & BEVERAGE
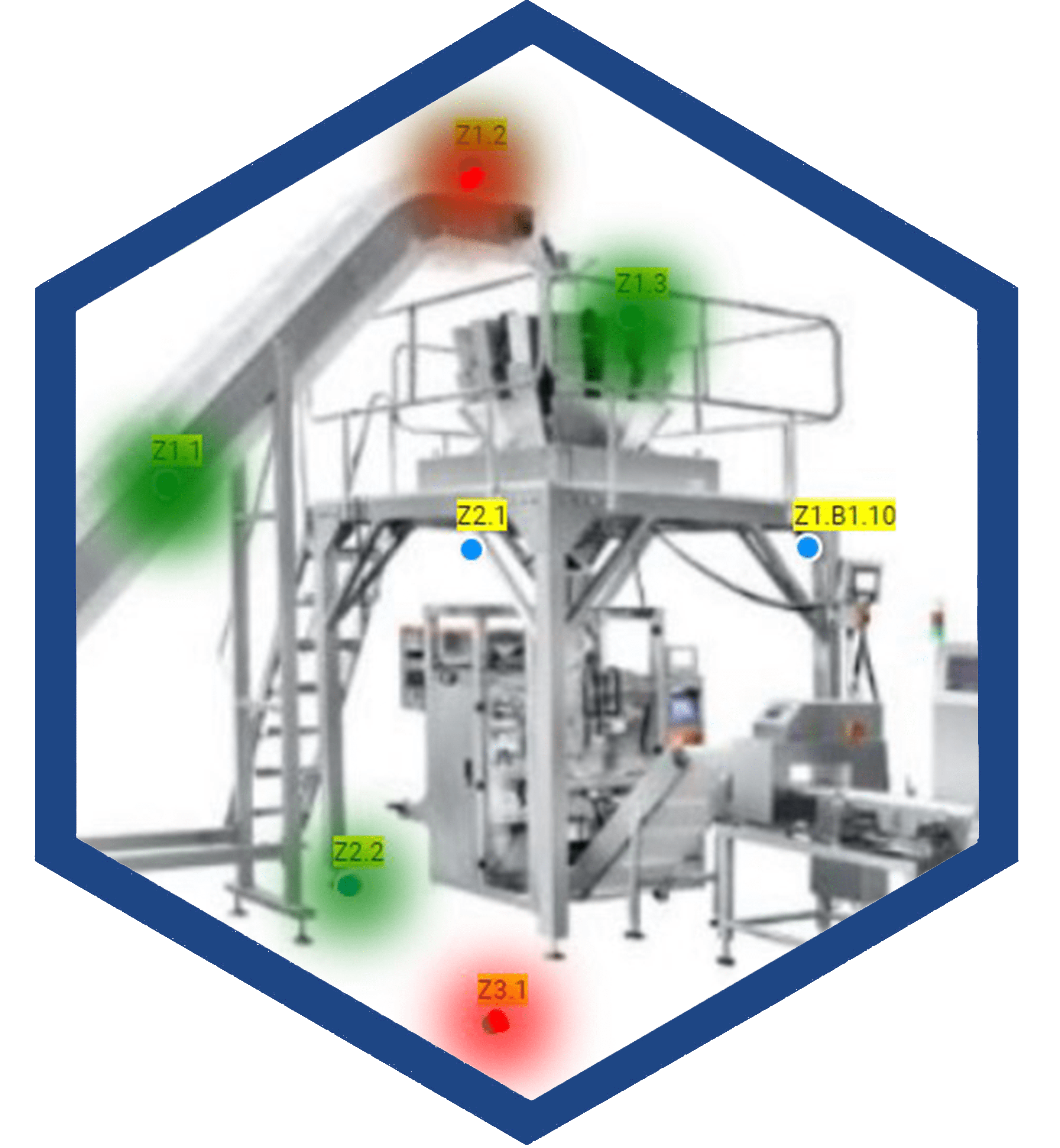
The global demand for food variety has grown, placing pressure on food suppliers at all levels. Differing regulations and standards make food safety and consistency a challenge that can be difficult to meet. In response to this need, Charm has developed a number of solutions specific to food suppliers. From Sanitation and Hygiene to Antibiotic/Mycotoxin, Pathogen and Allergen tests, we have the science-based test to give you—and your customers—peace of mind.
GRAINS
Global feed and grain challenges haven’t just hit human-products like dairy and food. Animal feed and grain has been impacted as well, leaving farmers everywhere working to address these challenges and regulations on their own. Charm helps meet this need with accurate, easy-to-perform, rapid tests for mycotoxins, antibiotics and pesticides—and more.
Meet Charm Sciences
The team at Charm Sciences has a vision—to apply science to deliver safer food to those who need it—and over the last 40 years, every one of our customers and employees play a key role in accomplishing this.
%
Today, over 75% of the global raw milk supply is tested with Charm products.
The U.S. Center for Disease Control reports that unpasteurized milk is 150 times more likely to cause foodborne maladies.
%
More than 50% of waterborne illnesses are caused by viral pathogens.
Approx. 48 million Americans get sick from contaminated food each year, resulting in 128,000 hospitalizations and 3,000 deaths.
“Our experience with the Charm products and organization have been outstanding. Their commitment to training our associates on the equipment during the start-up of the program and ongoing as new people have come to the organization has been second to none. They are extremely responsive to site/location needs for equipment and products and have been a key driver in the improvement of our Quality and Food Safety programs.”
QA Cheese Division | Fortune 100 Company
The First Test Approved for Simultaneous Detection of Three Antibiotic Families
Taking only 3 minutes, The Charm ROSA TRIO 2400 is the fastest 3-minute test approved for dairy milk antibiotic screening. Updated for Sulfonamide and Tetracycline drug family screening, ROSA was approved by the National Conference of Interstate Milk Shipments (NCIMS) and distributed by the Food and Drug Administration to state public health laboratories May 25, 2018.
TRIO is the first test approval ever for simultaneous detection of three antibiotic families, beta-lactams, sulfonamides and tetracyclines. TRIO represents the first time a single test result is able to meet both the mandatory and voluntary milk screening programs of the Pasteurized Milk Ordinance and NCIMS guidelines.
Subscribe to Our Newsletter for the Latest Industry News and Product Updates
Subscribe to Our Newsletter for the Latest Industry News and Product Updates
By submitting your information via this form, you give Charm Sciences permission to contact you via email about updates, news, or offers that may interest you. It is Charm Sciences’ policy not to share any personally-identifying material obtained through our websites with any third party.